Health & Fitness
Opioid Crisis: Virginia Sues Over High Prices of Treatment Drugs
The makers of Suboxone, a prescription drug used to treat opioid addiction, face a lawsuit from 36 states claiming prices were kept high.

RICHMOND, VA — Virginia and 35 other states have filed an antitrust lawsuit against the makers of Suboxone, a prescription drug used to treat opioid addiction, over allegations that the companies engaged in a scheme to block generic competitors and cause purchasers to pay artificially high prices.
The lawsuit, filed Thursday in the U.S. District Court for the Eastern District of Pennsylvania, accuses the companies of violating the federal Sherman Act and state laws. Counts include conspiracy to monopolize and illegal restraint of trade. In the suit, the attorneys general ask the court to stop the companies from engaging in anti-competitive conduct, to restore competition, and to order appropriate relief for consumers and the states, plus costs and fees.
"For many Virginians struggling with an addiction to heroin and other opioids, Suboxone can be an important part of a treatment plan that allows them to manage their substance abuse disorder," said Attorney General Mark Herring. "After extensive investigation, my colleagues and I have reason to believe that these monopoly practices violated the law and made this important medication more expensive and more difficult to obtain. I will continue to attack the heroin and prescription opioid crisis with every tool at my disposal and will not hesitate to take action against those who contribute to the problem, no matter who they are."
Find out what's happening in Dundalkfor free with the latest updates from Patch.
Reckitt Benckiser Pharmaceuticals, now known as Indivior, is accused of conspiring with MonoSol Rx to switch Suboxone from a tablet version to a film (that dissolves in the mouth) in order to prevent or delay generic alternatives from coming onto the market and to maintain monopoly profits.
The companies are accused of violating state and federal antitrust laws. As a result, the attorneys general claim that consumers and purchasers have paid artificially high monopoly prices, potentially since October 2009, when generic alternatives of Suboxone might otherwise have become available. During that time, annual sales of Suboxone topped $1 billion.
Find out what's happening in Dundalkfor free with the latest updates from Patch.
“Some people rely on this prescription drug to treat heroin addiction,” DeWine said. “They shouldn’t be forced to pay higher prices or deprived of options because drug makers circumvent the law to maximize their profits. People deserve the benefits of fair market competition. When the product involved is used to treat addiction, the implications are even more significant.”
Suboxone is a brand-name prescription drug used to treat opioid addictions by easing addiction cravings.
When Reckitt introduced Suboxone in 2002 (in tablet form), it had exclusive patent protection that lasted for seven years, meaning no generic version could enter the market during that time. The attorneys general allege, however, that before that period ended, Reckitt engaged in illegal “product hopping,” in which a company makes modest changes to its product to extend patent protections so other companies can’t enter the market and offer cheaper generic alternatives.
According to the lawsuit, Reckitt worked with MonoSol to create a new version of Suboxone – a dissolvable film, similar in size to a breath strip. Over time, Reckitt allegedly converted the market away from the tablet to the film through marketing, price adjustments and other methods. Ultimately, after the majority of Suboxone prescriptions were written for the film, Reckitt removed the tablet from the U.S. market. The complaint alleges that this was done to keep generic alternatives to Suboxone off the market and deprive consumers of the option of substituting a lower-cost generic alternative. (Currently, no generic alternative of the film is available.)
The lawsuit alleges that the Suboxone film provided no real benefit over the tablet and that Reckitt continued to sell the tablets in other countries even after removing them from the U.S. market. Reckitt also allegedly expressed unproven safety concerns about the tablet version and intentionally delayed FDA approval of generic versions of Suboxone.
Ohio was one of the lead states in the multi-state investigation that led to the lawsuit. The other states involved in the lawsuit are: Alabama, Alaska, Arkansas, California, Colorado, District of Columbia, Connecticut, Delaware, Florida, Hawaii, Illinois, Iowa, Kansas, Kentucky, Louisiana, Maine, Maryland, Massachusetts, Michigan, Minnesota, Mississippi, Missouri, Nebraska, New York, North Carolina, Oklahoma, Pennsylvania, Rhode Island, South Carolina, Tennessee, Utah, Vermont, Virginia, Washington, and Wisconsin.
— Patch Editor Chris Mosby reported this story.
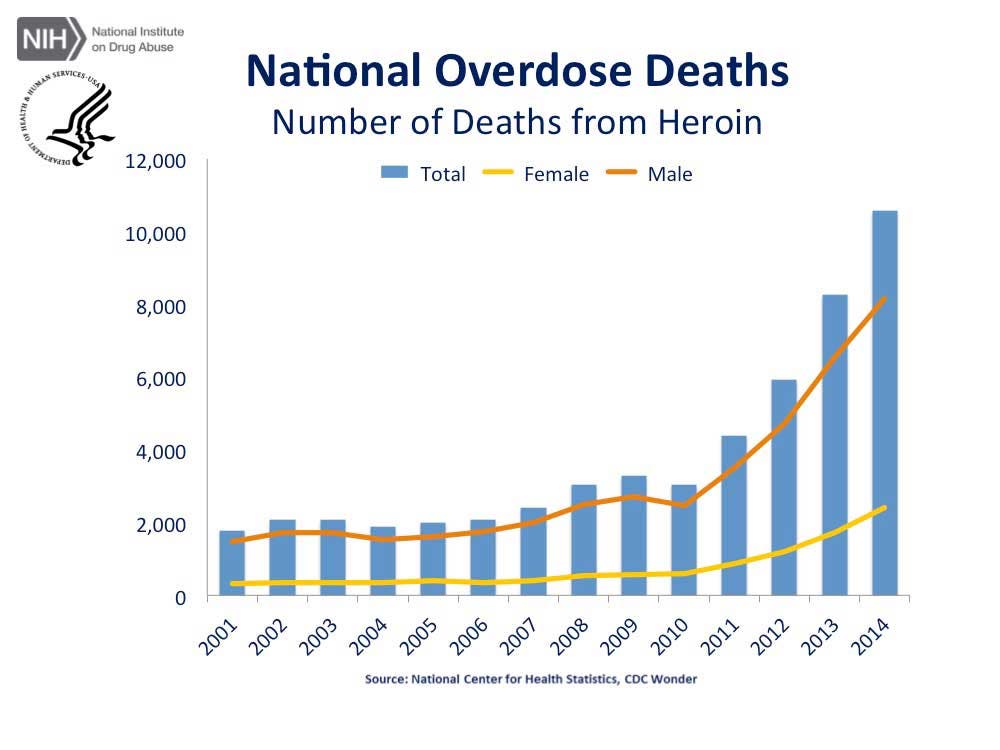
Image from Shutterstock, graphics from the National Institute of Health
Get more local news delivered straight to your inbox. Sign up for free Patch newsletters and alerts.