Health & Fitness
Infant Ibuprofen Sold In NJ Walmart, CVS Stores Is Recalled
A New Jersey-based company has recalled a medicine used as an infant pain reliever and fever reducer that's sold in various stores.
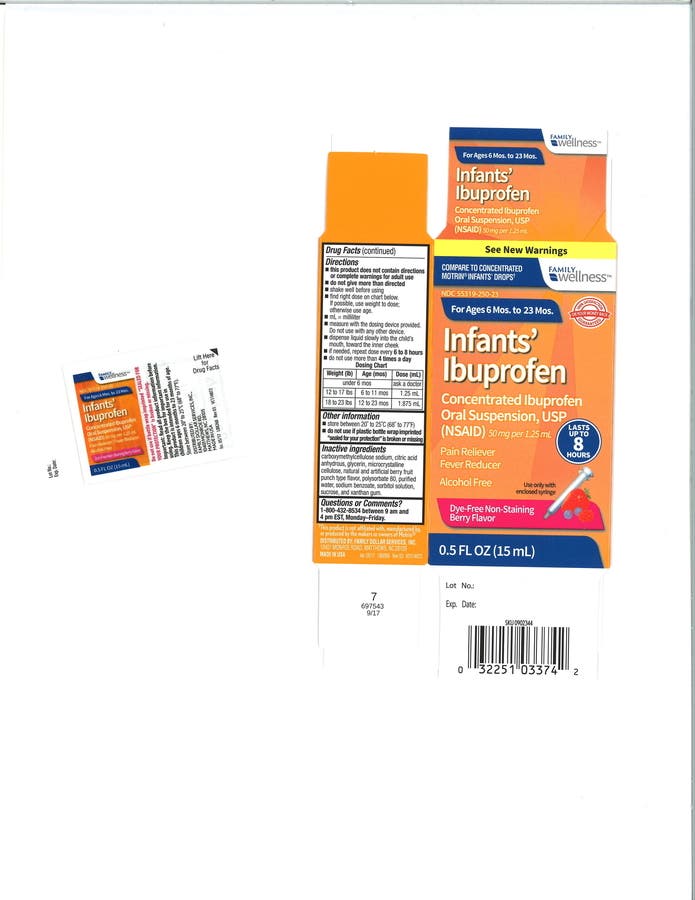
A New Jersey-based company this week has recalled a medicine used as an infant pain reliever and fever reducer that's sold in Walmart, CVS and other stores.
Tris Pharma, which is based in South Brunswick, has voluntarily recalled "Infants' Ibuprofen" because of potentially high levels of concentrations of the drug.
In a press release, company said there is a remote possibility that infants may be more susceptible to a higher potency level of drug, and therefore may be more vulnerable to permanent renal injury.
Find out what's happening in South Brunswickfor free with the latest updates from Patch.
Adverse effects that may be experienced are nausea, vomiting, epigastric pain or, more rarely, diarrhea. Tinnitus, headache and gastrointestinal bleeding are also possible adverse effects, according to the release.
To date, Tris Pharma, Inc. says it has not received any reports of adverse events related to the product that are the subject of this recall.
Find out what's happening in South Brunswickfor free with the latest updates from Patch.
The product was packaged in 0.5 oz. bottles for the recalled items listed below:
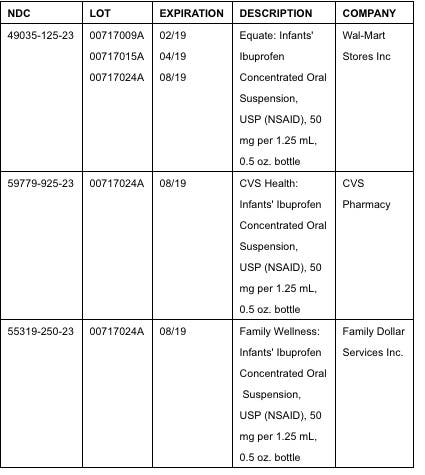
Tris Pharma, Inc. sold the affected product to one customer. The company has notified its customer by "urgent recall notice" and is arranging for the return of the recalled product, according to the release.
Consumers with questions regarding this recall can contact Tris Customer Service at 732-940-0358 (Monday through Friday, 8 a.m. ET through 5 p.m. PT) or via email at micc_tris@vigilarebp.com.
Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to taking or using this drug product.
Adverse reactions or other problems experienced with the use of the product lots subject to this recall may be reported to the FDA's MedWatch Adverse Event Reporting program either online, by regular mail or by fax: Download the form from www.fda.gov/MedWatch/getforms.htm or call 1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178
Tris Pharma photo
Get more local news delivered straight to your inbox. Sign up for free Patch newsletters and alerts.