Health & Fitness
New Cig Pack Pic Warnings Winner is Big Pharm
New cigarette pack warning pictures tell smokers to call 1-800-QUIT-NOW, a service that highly recommends the use of approved stop smoking products, when government evidence shows they don't work.
Yesterday the FDA approved nine frightening new cigarette pack warning pictures which must begin rotation and cover 50 percent of the face of all cigarette packs sold in the U.S. beginning in September 2012.
The common thread between all warnings is the message that smokers should call "1-800-QUIT-NOW." There, callers will be strongly advised to purchase pharmaceutical industry quitting products, products government health officials know do not prevail over unassisted cold turkey quitting.
SmokeFree.gov and 1-800-QUIT-NOW are the government's two primary stop-smoking resources. The quitting advice received when calling 1-800-QUIT NOW generally mirrors the advice at SmokeFree.gov.
Find out what's happening in Goose Creekfor free with the latest updates from Patch.
SmokeFree.gov should be renamed BuyMeds.now as smokers thinking about quitting are bombarded with 173 recommendations to purchase "medicine" or "medication."
While FDA approved quitting products generally clobber users of placebo look-a-likes inside randomized clinical trials (RCTs), placebo isn't a real quitting method.
Find out what's happening in Goose Creekfor free with the latest updates from Patch.
Nearly all real-world quitting method surveys to date, including the U.S. government's only known survey, show quitting products failing to prevail long-term over non-medication quitters.
Laughing all the way to the bank is GlaxoSmithKline, seller of Nicorette gum, the Nicoderm patch, the Commit nicotine lozenge, and the prescription pill Zyban.
Stock prices should jump at Norvitis, seller of the Habitrol nicotine patch, and for struggling Pfizer, maker of the Nicotrol inhaler and the controversial prescription pill Chantix - which has now recorded more than 10,000 reports of serious adverse events.
Smokefree.gov's "Medications to Help You Quit" page declares that "medicines can double your chances of quitting for good." Its "Quit Guide" parrots the claim that "medicines can double your chances of quitting for good."
Its "Dispelling Myths about Nicotine Replacement Therapy" page continues the drum roll that, "NRT can double a smoker's chances of quitting smoking."
And the chorus reaches crescendo inside the site's Clearing the Air quitting booklet which claims, "Studies show that these medications, compared with trying to quit without them, can double or triple your chances of quitting for good."
The problem is that randomized placebo-controlled clinical trials were never about "your" chances, as real cold turkey quitters who fully expect to meet, greet and move beyond full-blown withdrawal did not participate in randomized trials.
Instead, these products prevailed over smokers joining studies hoping to receive weeks or months of free quitting products, products they hoped would diminish their withdrawal syndrome.
Far from doubling "your" chances, a 2006 National Cancer Institute survey found that those quitting without medications actually had slightly higher long-term success rates than those using them.
Yes, as horrible as this sounds, our national "double your chances" health deception is undercutting national cessation efforts, contributing to relapse, and helping foster smoking related disease, disability and death.
In fact, since 2005, with rapidly rising cigarette prices, fewer places to legally smoke, scores of new smoking health study scares, and the biggest quitting meds marketing campaign in history, the adult U.S. smoking rate has remained stalled at 21 percent.
But they promise to double "your" chances? How can that be?
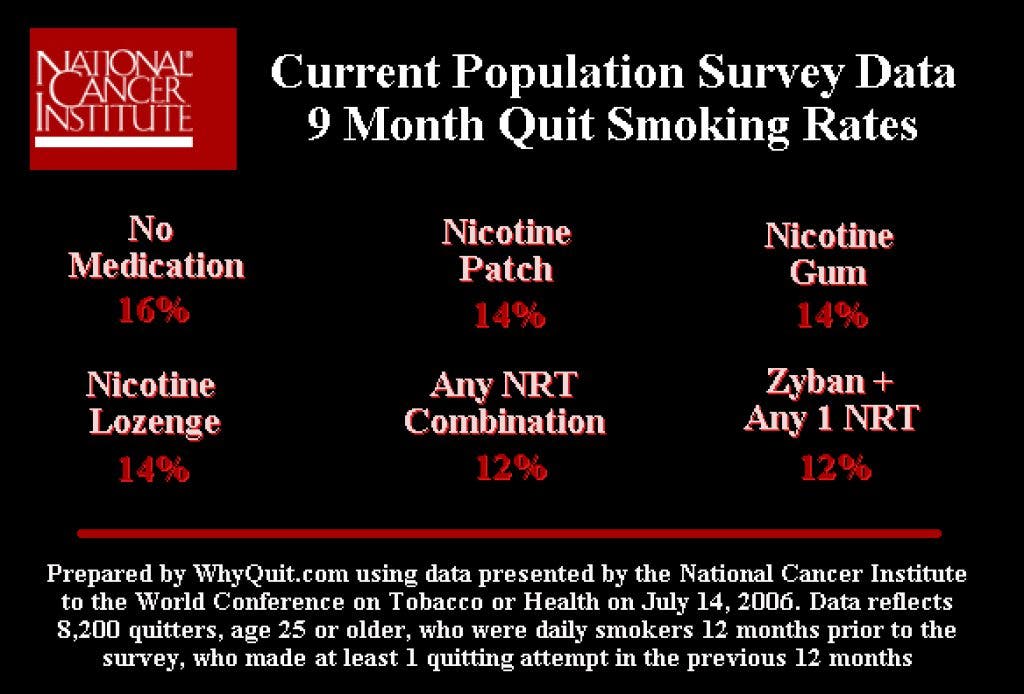
Pulitzer Prize winning journalist Kevin Helliker exposed the depth of pharmaceutical influence over U.S. cessation policy in a 2007 front page Wall Street Journal article entitled, "Nicotine Fix - Behind Antismoking Policy, Influence of Drug Industry."
The story shared unpublished data from a June 2006 NCI survey of 8,200 quitters. That study found that 14 percent quitting without medication were still not smoking after nine months, compared to only 12 percent among nicotine patch, gum and lozenge users.
Medication's dismal real-world failure is not new news. Results of the world's largest smoking cessation survey ever were published in the September 2002 edition of the Journal of the American Medical Association. It concluded that, "NRT appears no longer effective in increasing long-term successful cessation in California smokers."
A 2006 Australian study followed the smoking patients of 1,000 family practice physicians. Not only did cold turkey success rates trounce medication quitters, the study found that cold turkey accounted for 1,942 of 2,207 former smokers, a whopping 88 percent of all success stories.
But won't 1-800-QUIT-NOW telephone counseling cause medication use to achieve rates and victories similar to those seen in randomized placebo-controlled clinical trials? All credible evidence suggests otherwise.
The UK's national smoking treatment program offers what is likely the highest quality face-to-face local counseling and support cessation programs of any nation. There, the only long-term study found that those quitting without medication have a 67 percent higher one-year success rate than NRT quitters, and a 77 percent higher rate than Zyban quitters.
And short term UK NHS rates are not much better. Arguably, pharmaceutical industry influence over national cessation policy is even greater in the UK. There, the industry has somehow convinced the government to only follow and report one-month quitting rates when product use continues for 8 to 12 weeks before users attempt to stop using the product and adjust to natural brain dopamine pathway stimulation.
Even so, 2007-08 four-week national stop smoking program data reported a 55 percent quitting rate for non-medication quitters 49 percent for those using nicotine replacement therapy products (see 1st XLS doc - Table 4.5), 2008-09 data reports a draw at 48 percent (see 1st XLS doc - Table 4.5) and 2009-10 national 4-week data reports a 49 percent non-medication rate versus 47 percent for replacement nicotine quitters ( see 1st XLS doc - 1.16MB - see Table 4.4).
Recently, financially conflicted researchers have attempted to get smokers to purchase and use multiple quitting products at the same time, to start using them prior to quitting and to use them longer than previously recommended.
For example, 2010 headlines claimed that tripling the length of time the nicotine patch is used from 8 to 24 weeks "boosts" success rates. What news stories failed to mention is that the full text of the study reported that only 2 of 282 participants who wore the nicotine patch for 24 weeks were able to stop smoking for an entire year, an extremely dismal one-year continuous cessation rate of 0.7 percent.
So why do quitting products prevail over placebo inside clinical trials yet fall flat on their face in real-world use? A number of factors combine to hand clinical trial medicines unearned victories over placebo.
The common thread between nicotine replacement products, Zyban and Chantix is that each helps minimize withdrawal by continuing to chemically satisfy brain dopamine pathway wanting and desire. Pitting a product that provides continued stimulation against a placebo product which doesn't is clearly a profit-motivated apples to oranges comparison.
In fact, few studies tested to see if at study's end the user was actually able to successfully end chemical stimulation. It's important because we know that roughly 40 percent of replacement nicotine is today used by chronic long-term users of at least six months.
More troubling is the fact that most trials were designed and funded by the pharmaceutical industry and the focus and timing of their counseling and support was never designed with the placebo group's cessation needs in mind, but to foster successful transfer to the dopamine stimulation product being test.
Most troubling is the fact that the more experienced a quitter is, the more expert they become at at recognizing their withdrawal syndrome. Studies have shown that placebo-controlled trials are not blind as claimed, as 2-4 times as many within the placebo group are able to correctly identify their randomized assignment as guess wrong.
If you'd made six prior failed quitting attempts and joined a “medicine” study hoping for free replacement nicotine, would you have stuck around and allowed researchers to continue toying with you six months to a year if within 24 hours you recognized that you had been given an inert look-alike placebo instead? Neither did many of them.
We cannot blame a profit driven industry for designing clinical trials so as to vastly inflate the worth of worthless quitting products. What should deeply trouble all of us is that our government, whose own real-world findings are contrary to industry double “your” chances marketing, for whatever reason refuses to expose obvious industry shell games.
What would be interesting to know is how hard the pharmaceutical industry pushed to get the 1-800-QUIT-NOW message on all cigarette packs, and the full extent of its financial influence in designing 1-800-QUIT-NOW telephone counseling scripts.
What can be declared with 100 percent certainty is that more long-term successful ex-smokers will succeed in quitting cold turkey this year than by all other quitting methods combined. Search as you might, it's a natural cessation message that our government refuses to share.