Community Corner
Public Works Facility Eyed As COVID-19 Vaccination Site
The McHenry County Department of Health could use the village of Algonquin's public works facility for distributing vaccines.

ALGONQUIN, IL — The Algonquin Public Works facility could serve as a site for distributing COVID-19 vaccines. On Tuesday, the Algonquin Village Board discussed an intergovernmental agreement with the McHenry County Department of Health for use of the facility and property at 100 Meyer Drive as a mass vaccination site.
The facility was picked for its size; its indoor garage space — which could be helpful during inclement weather and for funneling cars through — and because of experience village staff has with emergency responses and traffic control.
Details surrounding the hours of service, final site logistics and staffing will be determined by staff and also be contingent upon when the approved vaccine is released, Bob Mitchard, public works director for the village of Algonquin, wrote in a memo to the village board.
Find out what's happening in Algonquin-Lake In The Hillsfor free with the latest updates from Patch.
The final site plan and distribution details will be approved by the village’s public works director, police chief, fire chief, and representatives of the McHenry County Department of Health. And more details would be then passed onto the village board at a later date, according to village documents.
Issues surrounding traffic flow and backups in the area were discussed during the village's Committee of a Whole meeting Tuesday. A nearby gravel road could possibly be used to funnel traffic through but the village would need to get approval from the business that owns that property, Mitchard said during the Tuesday meeting.
Find out what's happening in Algonquin-Lake In The Hillsfor free with the latest updates from Patch.
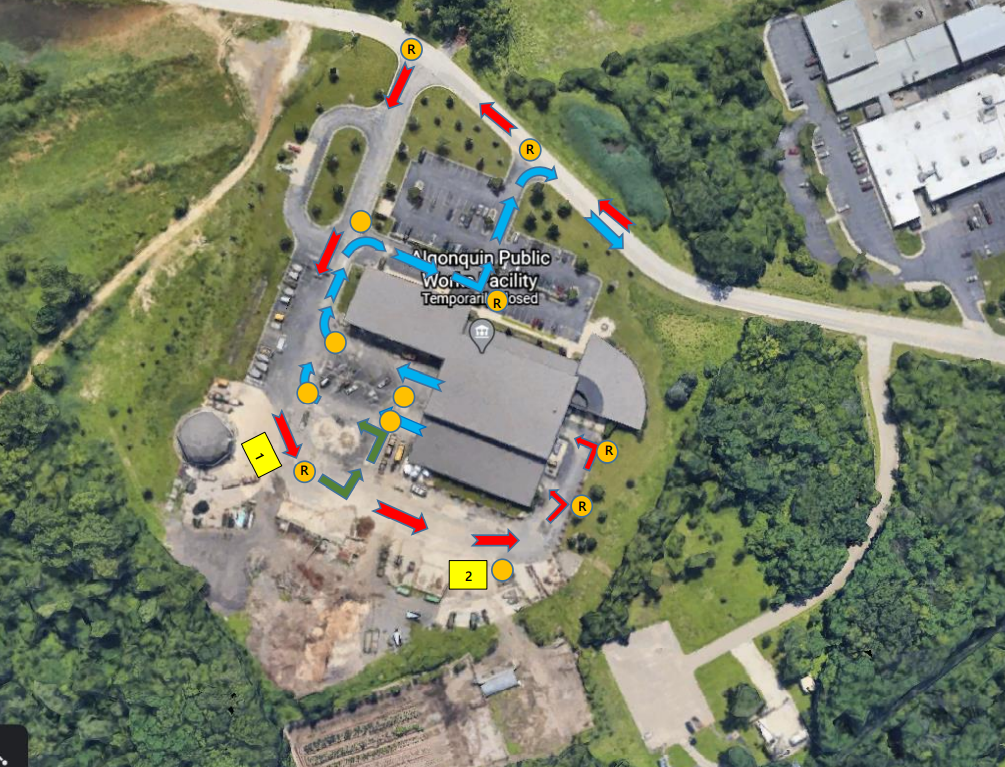
The term of the memorandum would run from November 2020 through Dec. 1, 2021.
Last week, Pfizer announced its vaccine was found to be 90 percent effective in preventing COVID-19. On Monday, Moderna Inc. announced an analysis of a clinical trial found its experimental vaccine was 94.5 percent effective in preventing the virus, according to NPR.
Get more local news delivered straight to your inbox. Sign up for free Patch newsletters and alerts.