Health & Fitness
Coronavirus Rapid Test For Screening Asymptomatic Questioned
Public health officials warn of limited data guiding the use of antigen testing — like the 150 million Abbott Labs tests purchased by HHS.

GREEN OAKS, IL — Rapid coronavirus tests like the Abbott Labs system used at the White House have a high risk of providing false positive results — or missing actual infections — especially when administered on people without symptoms or close contacts of someone with COVID-19, federal regulators warned.
In a letter to health care providers and staff at clinical laboratories, U.S. Food and Drug Administration last week said labs should expect some false positives when screening large populations with relatively few active infections.
"As disease prevalence decreases, the percent of test results that are false positives increase," according to the FDA.
Find out what's happening in Lake Forest-Lake Blufffor free with the latest updates from Patch.
Antigen testing is a faster and cheaper form of viral testing than the gold standard real-time reverse transcriptase polymerase chain reaction, or RT-PCR, tests.
"The FDA is aware of reports of false positive results associated with antigen tests used in nursing homes and other settings and continues to monitor and evaluate these reports and other available information about device safety and performance," the administration's letter said.
Find out what's happening in Lake Forest-Lake Blufffor free with the latest updates from Patch.
According to guidance from the Illinois Department of Public Health, follow-up testing is recommended if the results from the antigen tests do not align with a patient's symptoms or likelihood of exposure.
The U.S. Department of Health and Human Services purchased about 150 million of Abbott's BinaxNOW antigen tests in a $760 million contract with the Green Oaks-based medical device maker announced in August.
“Over the past months, our collaboration with Abbott has exemplified the Administration’s whole-of-society approach to this pandemic, fully leveraging America’s industry and innovative spirit,” said Dr. Brett Giroir, the Health and Human Services official in charge of the federal government's COVID-19 diagnostic testing efforts.
“Under President Trump’s leadership we have worked closely with the private sector to stimulate testing innovations such as the Abbott BinaxNOW COVID-19 test," Giroir added in a statement announcing the contract. This is no small achievement and is of an unprecedented scale. The Administration is now prepared to fully deploy this new testing asset to continue combatting the virus and safely and sensibly reopening our great nation.”
Related:
Abbott's New Coronavirus Antibody Test Detects Recent Exposures
Abbott Ships Over Rapid COVID-19 Tests To States
Emergency Authorization Given To Rapid Coronavirus Test
Abbott's rapid COVID-19 card tests uses nasal swabs and can be completed in minutes at a cost of about $5 per use. According to the FDA, Abbott was the only known source to scale up and immediately provide the needed testing supplies.
In addition to their use screening visitors to the White House — including before a so-called "superspreader" event on Sept. 26 — hundreds of thousands of the rapid tests have been sent out to state governments, such as Louisiana. Public health officials there have received more than 300,000 tests and expect to be sent 1.4 million more, but updated testing guidance issued last week does not recommend they be used to screen people without COVID-19 symptoms or those who have come into contact with someone who has the virus.
"At this time," according to the guidance from the Louisiana Department of Public Health, "LDH does not recommend antigen tests for screening of asymptomatic individuals with no known exposure to a person diagnosed with COVID-19." Bloomberg reported public health authorities in Oregon also recommended against the use of BinaxNOW for screening asymptomatic people, and Nevada last month instructed nursing homes to stop using the tests before reversing course.
In Illinois, guidance from state public health officials said rapid antigen tests for screening at the point of care, or POC, are most appropriate in high-risk congregate settings to quickly identify people with infections. But they also have a higher chance of failing to detect an active infection.
"Therefore, it may be necessary to confirm a rapid POC antigen or rapid POC molecular test result with a RT-PCR test, especially if the result of the rapid POC test is inconsistent with the clinical perspective, i.e., a negative antigen test on a symptomatic individual or on a person who is a close contact to a confirmed or probable case," according to a letter to health care providers last month from Dr. Catherine Counard, medical officer of the Office of Health Protection in the Illinois Department of Public Health.
"Rapid antigen tests perform best when the person is tested in the early stages of infection with SARS-CoV-2 when viral load is generally highest. They also may be informative in diagnostic testing situations in which the person has a known exposure to a confirmed or probable case of COVID-19."
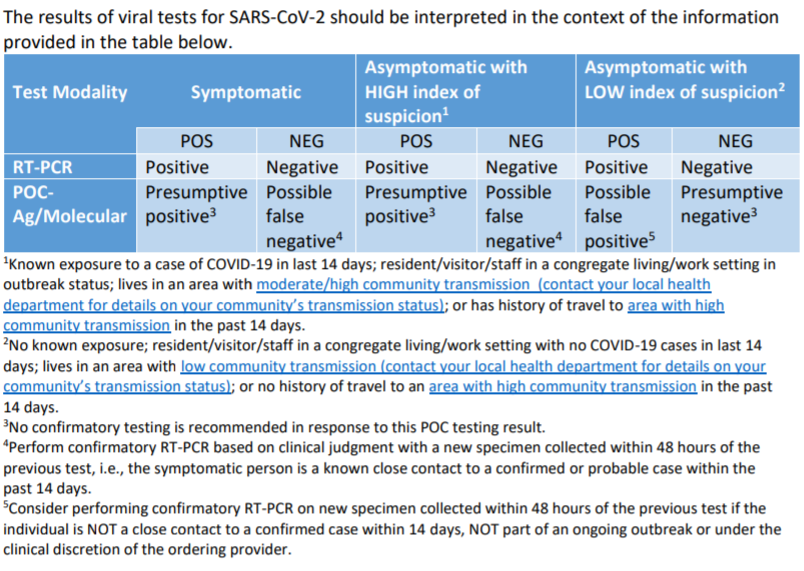
According to Counard's testing recommendations, confirmatory PCR tests must take place within two days of an exposure and without chances for new exposures in between the initial test result and confirmation.
In guidance to nursing homes, the medical officer noted there "are only limited data to guide the use of rapid antigen tests to screen asymptomatic persons to detect COVID-19 infection."
On Oct. 14, Illinois public health officials began adding the numbers of antigen testing completed to state totals.
Last week, Giroir, the HHS official coordinating administration's federal diagnostic efforts, told reporters antigen testing was the "only way" to screen millions of people for coronavirus every month.
Get more local news delivered straight to your inbox. Sign up for free Patch newsletters and alerts.