Health & Fitness
Scripps Health Using Plasma To Treat Coronavirus Patients
Scripps Health became the first health care provider in the county to use an experimental plasma therapy, the health care system announced.
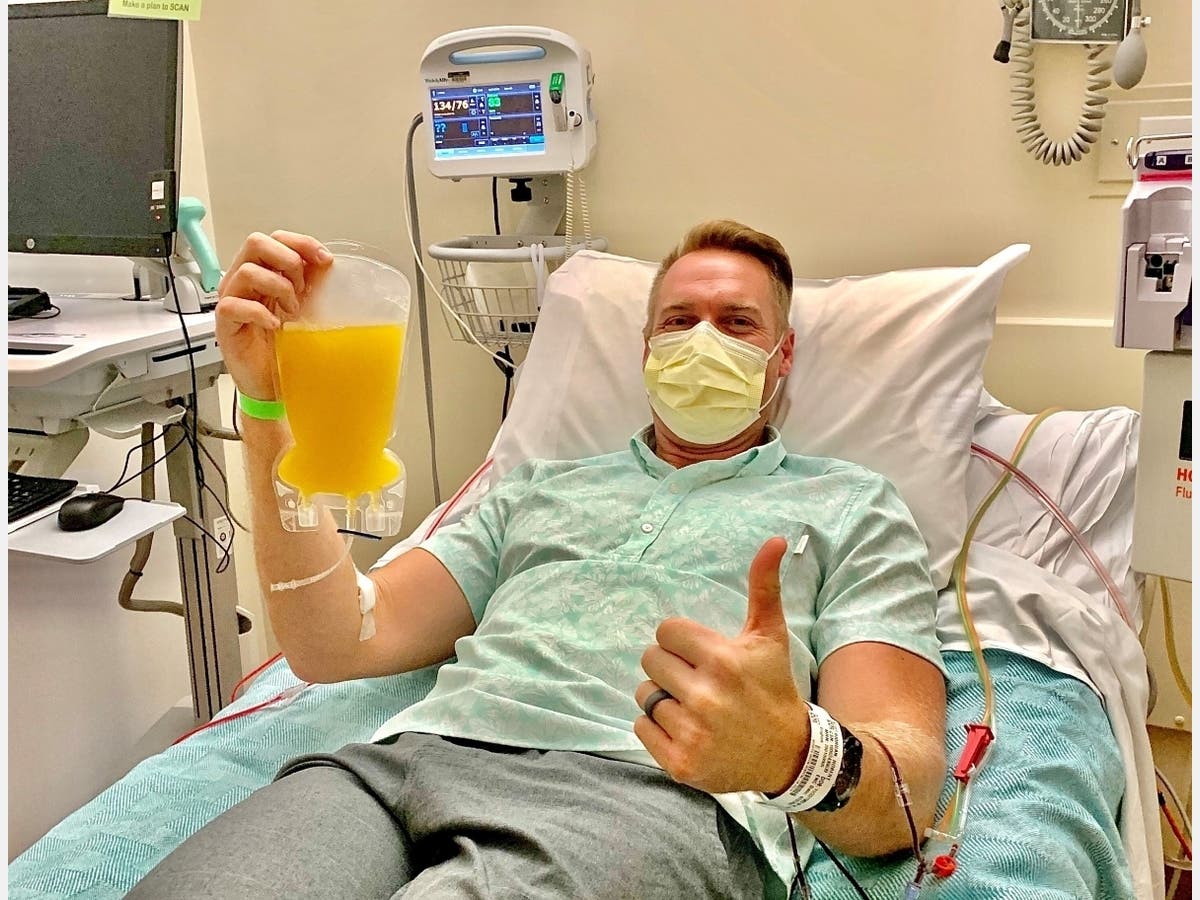
SAN DIEGO, CA — Scripps Health recently became the first health care provider in San Diego County to use an experimental plasma therapy as a possible treatment for COVID-19 patients, the health care system announced Friday.
The treatment involves transfusing plasma donated by someone who has recovered from COVID-19 into a hospitalized patient who is battling a serious COVID-19 infection. Plasma is the almost-clear liquid that remains after red and white blood cells and platelets are removed from the blood.
The idea behind the treatment — known as convalescent plasma therapy — is that people who have fully recovered from COVID-19 have antibodies in their plasma that can attack the virus when transfused into patients with serious, active disease.
Find out what's happening in San Diegofor free with the latest updates from Patch.
Don't miss local and statewide news about coronavirus developments and precautions. Sign up for Patch news alerts and newsletters.
On Wednesday, Encinitas resident Robert Riordan visited the Scripps Blood and Marrow Transplant Program at Scripps Green Hospital to donate plasma. Riordan was diagnosed with COVID-19 on March 19 after a ski trip to Colorado, according to Scripps Health. He has been symptom-free since March 27.
Find out what's happening in San Diegofor free with the latest updates from Patch.
Riordan is the second person who donated convalescent plasma at Scripps, which will be transfused into a patient being treated in the intensive care unit at Scripps Green. The first convalescent plasma collection and transfusion was conducted at Scripps Green with a different donor and patient on April 1.
While the therapy is still experimental, the U.S. Food and Drug Administration on March 24 allowed doctors to use plasma from recovered patients to treat those with "serious or immediately life-threatening COVID-19 infections" under an emergency approval system. Doctors can apply to the FDA to use it for their patients, and the agency will review the requests quickly and make decisions on a case-by-case basis.
The first convalescent plasma treatments in the U.S. for COVID-19 were performed in late March in New York and Texas, and other centers throughout the country are now adopting the therapy. Doctors and researchers will be monitoring progress closely, as they know it will take time to determine how well convalescent plasma works against COVID-19.
The strategy of transfusing convalescent plasma has been used in the past to treat viral disease outbreaks of polio, measles and mumps before a vaccine was available. More recently it has been used with some effectiveness to treat patients with SARS and Ebola.
Also see:
- California Coronavirus: Latest Updates On Cases, Orders, Closures
- Coronavirus In San Diego County: Latest News
- San Diego Residents Urged To 'Stay The Course' Following Protests
- 7 More Deaths As Coronavirus Cases Surpass 2,000 In San Diego
- Coronavirus Cases Surpass 1,000 Milestone In San Diego County
- Face Coverings Urged As Coronavirus Cases Rise In San Diego
- San Diego Authorities To Crack Down On Stay-At-Home Violators
Get more local news delivered straight to your inbox. Sign up for free Patch newsletters and alerts.