Community Corner
Experimental Ebola Vaccine Shows Promise: Researchers
The vaccine is being developed and tested at the National Institutes of Health in Bethesda.
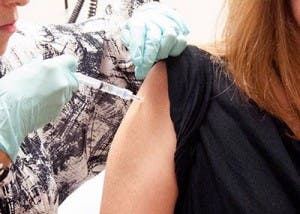
An experimental Ebola virus vaccine tested at the National Institutes of Health in Bethesda shows promising results, and has medical researchers planning to accelerate the pace of human trials.
The vaccine to prevent Ebola virus disease produced antibodies in all 20 healthy adults who received it in a clinical trial. Many of the test subjects also had a T-cell response to the injection, which medical authorities say is the key to protecting people from the disease when they are later exposed to Ebola.
Two American health-care workers were treated for Ebola in a special isolation unit at the Bethesda campus earlier this fall. Both recovered from the illness.
Find out what's happening in Bethesda-Chevy Chasefor free with the latest updates from Patch.
The first was a doctor who was exposed to the Ebola virus while volunteering with patients in Sierra Leone; he arrived at the NIH Clinical Center Sept. 28, Patch earlier reported.
Then Texas nurse Nina Pham was flown to Bethesda in mid-October after she contracted the virus while caring for Ebola patient Thomas Eric Duncan at Texas Health Presbyterian Hospital in Dallas. Duncan died Oct. 8 from the infectious disease.
Find out what's happening in Bethesda-Chevy Chasefor free with the latest updates from Patch.
The experimental vaccine being developed in response to the Ebola outbreak in West Africa does not contain the Ebola virus and cannot cause the often-deadly disease, says the NIH. All the clinical trial volunteers developed antibodies to fight the virus within four weeks of receiving the vaccine.
The vaccine undergoing tests was co-developed by the NIH’s National Institute of Allergy and Infectious Diseases and drug company GlaxoSmithKline, and was tested at the NIH Clinical Center in Bethesda.
“The unprecedented scale of the current Ebola outbreak in West Africa has intensified efforts to develop safe and effective vaccines, which may play a role in bringing this epidemic to an end and undoubtedly will be critically important in preventing future large outbreaks,” said NIAID Director Anthony S. Fauci, M.D. “Based on these positive results from the first human trial of this candidate vaccine, we are continuing our accelerated plan for larger trials to determine if the vaccine is efficacious in preventing Ebola infection.”
The National Institutes of Health in Bethesda is one of four facilities in the United States with special isolation facilities where Ebola patients can more easily be treated. Other specialized facilities are Emory University Hospital in Atlanta; Nebraska Medical Center in Omaha; and St. Patrick Hospital in Missoula, MT.
More information about the clinical trials will be published in the New England Journal of Medicine.
»A woman enrolled in the first test of an experimental Ebola vaccine receives a dose of the vaccine at the NIH Clinical Center in Bethesda on Sept. 2. Photo from the National Institutes of Health
Get more local news delivered straight to your inbox. Sign up for free Patch newsletters and alerts.