Health & Fitness
FDA Authorizes 2nd Booster: What To Know In VA, DC
Eligible Virginia, DC residents can get a 2nd Pfizer or Moderna COVID booster at least 4 months after getting the first booster, FDA said.
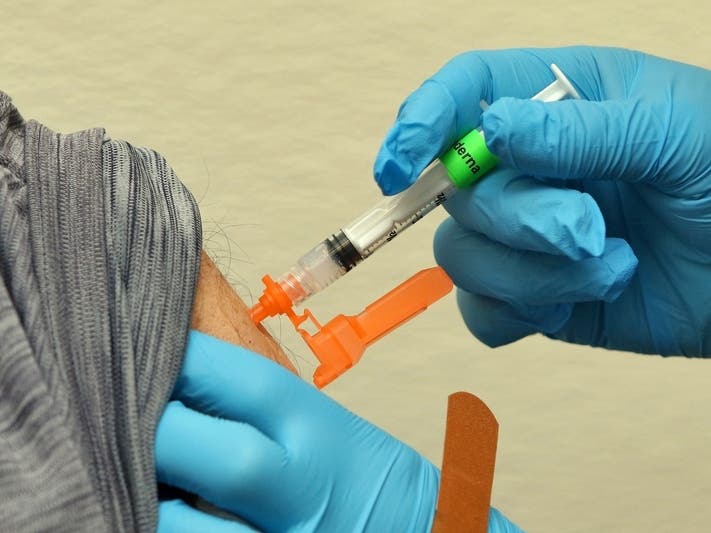
VIRGINIA — With the number of COVID-19 cases declining in Virginia, there is more good news in the fight against the disease. People 50 and older and anyone who is immunocompromised in Virginia and DC may get a second COVID-19 booster shot, the Food and Drug Administration said Tuesday.
In the District, COVID rates have increased in the past two weeks, on average by 20 percent or 81 new cases per day, according to the New York Times.
Those eligible can receive either the Pfizer or Moderna booster at least four months after getting the first booster from either vaccine manufacturer. For those 12 and older who are immunocompromised, the FDA authorized a second Pfizer booster at least four months after the initial booster. The Moderna booster may be given to those 18 and older who are immunocompromised after the same period.
Find out what's happening in Across Virginiafor free with the latest updates from Patch.
In its announcement, the FDA cited evidence of waning immunity in older and immunocompromised people, and it urged anyone who has not gotten their initial booster dose to do so.
“Based on an analysis of emerging data, a second booster dose of either the Pfizer-BioNTech or Moderna COVID-19 vaccine could help increase protection levels for these higher-risk individuals,” said Dr. Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research. “Additionally, the data show that an initial booster dose is critical in helping to protect all adults from the potentially severe outcomes of COVID-19. So, those who have not received their initial booster dose are strongly encouraged to do so.”
Find out what's happening in Across Virginiafor free with the latest updates from Patch.
The agency said the benefits of the second booster outweigh any risks among these groups. For anyone else, the authorization of the first booster dose remains unchanged.
COVID-19 case numbers, hospitalizations and deaths are declining nationwide, but health officials are keeping an eye on the worrying omicron subvariant, BA.2, or “stealth omicron,” which has caused new cases to more than double in the United Kingdom in recent weeks, National Public Radio reported.
Virginia has one of the country's lowest coronavirus rates, with eight cases per 100,000 residents in the state, according to the New York Times COVID tracker. The state's infection rate has plunged 26 percent in the past two weeks with an average of 705 new cases per day.
Virginia's COVID dashboard puts the positivity count at 3.2 percent, with 867 new cases in the past 24 hours.
In total, the state has confirmed 1,667,289 cases during the pandemic.
Just past the two-year mark of the pandemic, Virginia has recorded a total of 19,653 deaths.
On average, Virginia has seen 397 residents hospitalized for COVID-19 in the last two weeks, a decrease of 31 percent.
Seventh-three percent of Virginians are fully vaccinated.
Approval of a second booster is a complicated matter, though, because health experts don’t know the timing of the next COVID-19 surge, The Associated Press reported. It is more difficult to predict for a number of reasons, including the use of at-home tests, whose results aren't included in government data; and the government’s shift in data collection to focus on hospitalizations, which occur after a surge has arrived.
Citing real-world data from Israel, among a handful of countries that offer a second booster shot, Pfizer asked the FDA to greenlight the additional booster shot for people 65 and older. The FDA also cited data from Israel, saying the second Pfizer booster revealed no new safety concerns.
Moderna followed suit, asking the FDA to approve a broad emergency use application of an additional vaccine booster to include any adult over 18 who has gotten any authorized or approved vaccine.
For the second Moderna booster, the agency said the decision was informed by experience with the Pfizer shot and data from a study that showed no new safety concerns up to three weeks after the second booster was administered.
Both vaccine makers cited studies that confirmed infections were two times lower and rates of severe illness were four times lower among people who received the second Pfizer booster, compared to people who received only one, according to a news release.
The administration didn’t need the blessing of either the FDA or Centers for Disease Control and Prevention to move forward, but Dr. Jesse L. Goodman, a former chief scientist at the FDA, had said skipping the advisory committees would be a mistake.
“This is a complex decision that involves a pretty deep dive, and I think it would really benefit from public discussion,” Goodman, a former chief scientist at the FDA, told the Times. “I would not want to see an advisory committee skipped on this.”
The FDA will convene its outside advisory committee in April to discuss whether COVID-19 booster shots should be part of the annual influenza vaccine campaign encouraging all Americans to get their shots in the fall, ahead of cold-weather season when people are cooped up inside, The Washington Post reported.
Get more local news delivered straight to your inbox. Sign up for free Patch newsletters and alerts.